
The aim of this brief review is to summarize the recent literature on the effect of ozone (O3) on cutaneous tissues. Recently it has been reported that a chronic contact with O3 can be deleterious for the skin. G. Valacchi, V. Fortino and V. Bocci group and others have shown a progressive depletion of antioxidant content in the stratum corneum and this can then lead to a cascade of effects resulting in an active cellular response in the deeper layers of the skin. Using an in vivo model we have shown an increase of proliferative, adaptive and proinflammatory cutaneous tissue responses. On the other hand the well known activity of O3 as a potent disinfectant and oxygen (O2) donor has been also studied for therapeutic use. Two approaches have been described. The first consists of a quasi-total body exposure in a thermostatically controlled cabin. This treatment has proved to be useful in patients with chronic limb ischaemia. The second approach is based on the topical application of ozonated olive oil in several kinds of skin infection (from soreness to diabetic ulcers, burns, traumatic and surgical wounds, abscesses and skin reactions after radiotherapy). G. Valacchi, V. Fortino and V. Bocci and other authors have observed a striking cleansing effect with improved oxygenation and enhanced healing of these conditions. It is now clear that, on the skin, O3, like other drugs, poisons and radiation, can display either a damaging effect from a long exposure or a beneficial effect after a brief exposure to O2 and O3 or to the application of ozonated oil to chronic wounds.
Christian Friedrich Schonbein discovered ozone (O3) in 1839 and in 1853 he made the first measurement of O3 in the Austrian mountains. Today, we know that some gases such as O3, carbon monoxide, nitric oxide and carbon dioxide can have dual actions, behaving either as useful or as harmful agents(1). The O3 layer is located at an altitude of about 22 km. Approximately 90% of the O3 in the atmosphere resides in the stratosphere. The O3 concentration in this region is about 10 parts per million by volume. O3 absorbs the bulk of solar ultraviolet (UV) radiation in the wavelengths from 290 to 320 nm. These wavelengths are harmful to life because they can be absorbed by the nucleic acid in cells and damage it. Increased penetration of UV radiation to the planet’s surface would damage plant life and have harmful environmental consequences. Appreciably increased amounts of solar UV radiation at the Earth’s surface would result in a host of biological effects, such as a dramatic increase in cancer; it seems that a 10% drop in the level of the O3 layer may cause a 25% increase in skin carcinoma and melanoma(2). Moreover this risk has recently been enhanced by excessive pollution with O3 in the troposphere, particularly evident during summertime in large cities(3). Thus the strong oxidative power of O3 in
association with other contaminants, can be harmful for plants and animals. The human bronchopulmonary system and the skin are the most accessible targets; they are vulnerable owing to the paucity of local antioxidant defences. O3 toxicity for the pulmonary system has been extensively examined while attention to the skin problem is more recent but no less important. An interesting difference that we would like to point out here is that while the pulmonary system is absolutely intolerant to O3 and this gas should never be inhaled, the skin, for anatomical and biochemical reasons, is somewhat more resistant.
Recent literature points out that although a long exposure is certainly deleterious, transitory exposure at low and precisely controlled O3 concentrations can have useful effects. The damage to the respiratory tract by oxidative environmental pollutants such as O3 and nitrogen oxides have already been reviewed(4) while recent literature has focused only on the damaging interaction between long exposures to O3 and cutaneous tissues(5–10). We believe that it is now also correct to discuss the unexpected therapeutic effect of a brief exposure of patients to O3 or the use of ozonated oil for cutaneous infections.
Skin responses to environmental stress
The skin consists of two main layers, the epidermis and the dermis, of which the latter is superficial to the subcutaneous fat tissue. Dermal fibroblasts synthesize a complex extracellular matrix containing collagenous and elastic fibres. Blood capillaries reach the upper part of the dermis. The epidermis contains mostly keratinocytes that rise to the skin surface as they differentiate progressively to form the non-nucleated corneocytes that comprise the superficial part of the epidermis, the stratum corneum (SC). The skin, as an interface between the body and the environment, is chronically exposed to stress from both UV radiation and environmental oxidative pollutants such as diesel fuel exhaust, cigarette smoke, halogenated hydrocarbons, heavy metals and O3 (one of the most toxic of these compounds)(11) The skin is protected against oxidative stress by a variety of antioxidants; these include enzymatic antioxidants such as glutathione peroxidase, superoxide dismutase, catalases and nonenzymatic low-molecular weight antioxidants such as vitamin E isoforms, vitamin C, glutathione (GSH), uric acid and ubiquinol(7).
Recently, the presence of a- and c-tocopherol, ascorbate, urate and GSH has been shown also in the SC(10). Interestingly, the distribution of antioxidants in the SC follows a gradient with higher concentrations in deeper layers(12) This may be explained by the fact that SC layers move up in time as a part of the physiological turnover of skin cells and are replaced by freshly differentiated keratinocytes. Therefore, the superficial layer is exposed to chronic oxidative stress for a longer time than the deep layer. Compared with the SC, the surface lipids contain high levels of a- and c-tocopherol because of the secretion of vitamin E by sebaceous glands(13) Eventually, the uppermost layer of the SC will desquamate and the remaining antioxidants and reacted products will be eliminated from the body. In general the outermost part of the skin, the epidermis, contains lower concentrations of antioxidants than the dermis. In the lipophilic phase, a-tocopherol is the most prominent antioxidant, while vitamin C and GSH are the most abundant in the aqueous phase.

Skin responses to ozone
It is generally understood that the toxic effects of O3 are mediated through free radical reactions, although O3 is not a radical species per se(14) They are achieved either directly by the oxidation of biomolecules to give classical radical species (hydroxyl radical) or by driving the radical-dependent production of cytotoxic, nonradical species (aldehydes)(15) Furthermore, the formation of the oxidation products characteristic of damage from free radicals has been shown to be prevented by the addition of the antioxidants vitamin E and C, though the mechanism is not fully understood. The target specificity of O3 towards specific compounds together with its physicochemical properties of fairly low aqueous solubility and diffusibility, must be taken into account when a target tissue (lung and skin) is exposed to O3(16).
Polyunsaturated fatty acids
Cell membranes and their lipids are relevant potential targets of environmental stressors such as UV and O3. Using a spin trapping technique, the formation of radicals in the SC upon exposure to O3 and⁄or UV was detected (L. Packer, unpublished observation). The spin adduct could arise from an alkoxyl radical formed during lipid peroxidation. Furthermore, lipid radicals (LÆ) are generated in epidermal homogenates that have been exposed to environmental stressors. The organic free radical LÆ reacts with O2, forming peroxyl radical LOOÆ and hydrolipoperoxides (LOOH). Transition metals and in particular iron, play a key role in the reactions of LOOH and in the subsequent generation of alkoxyl radicals (ROÆ can amplify the lipid peroxidation process).
The stratum corneum as the first target of environmental stressors
Within the skin, the SC has been identified as the main target of oxidative damage(17,18). As the outer skin barrier, the SC has important functions, limiting transepidermal water loss and posing a mechanical barrier to penetration by exogenous chemicals and pathogens. It comprises a unique two-compartment system of structural, non-nucleated cells (corneocytes) embedded in a lipid enriched intercellular matrix, forming stacks of bilayers that are rich in ceramides, cholesterol and free fatty acids.(19,20)
The effects of O3 on cutaneous tissues have recently been evaluated using a murine model. While no effect of O3 on endogenous antioxidants was observed in full thickness skin (dermis, epidermis and SC), it could be demonstrated that a single high dose of O3
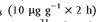
significantly depleted topically applied vitamin E.(21) When the skin was separated into upper epidermis, lower epidermis and papillary dermis, and dermis, O3 induced a significant depletion of tocopherols and ascorbate followed by an increase in the lipid peroxidation measured as malondialdehyde (MDA) content. O3 is known to react readily with biomolecules and does not penetrate through the cells; therefore, it was hypothesized that O3 mainly reacts within the SC.(17) This hypothesis was supported by further experiments, where hairless mice were exposed to varying levels of O3 for 2 h. Depletion of SC lipophilic (tocopherols) as well as hydrophilic (ascorbate, urate, GSH) antioxidants was detected upon O3 exposure and it was accompanied by a rise in lipid peroxidation as an indicator of increased oxidative stress.(22) Furthermore, a recent study has shown the increase of 4-hydroxylnonenal (4-HNE) content in murine SC using both Western blot and immunohistochemical analysis.(23)
Skin cellular responses to ozone exposure
As mentioned above, O3 exposure was shown to induce antioxidant depletion as well as lipid and protein oxidation in the SC. Recent studies have investigated the effects of O3 in the deeper functional layers of the skin.(23–25)
To evaluate the effect on cutaneous tissues of O3 exposure, hairless mice were exposed for 6 days to

)1 for 6 h day)1 and the homogenized whole skin was analysed. Under these experimental conditions an increase of proinflammatory marker cyclooxygenase-2 (COX-2) expression was detected confirming the role that O3 can play in skin inflammation. This induction was accompanied by an increase in the protein level of heat shock protein (HSP)(32), also known as haem oxygenase-1 (HO-1), confirming that HSPs are sensitive markers of O3-induced stress in cutaneous tissues. Our group was the first to document the upregulation of HSPs 27, 32 and 70 in homogenized murine skin upon O3 exposure

HSP27 showed the earliest (2 h) and highest (20-fold) response to O3 compared with the delayed induction (12 h) of HSP70 and HO-1. Increased expression of HSP27 has been demonstrated following heating of both keratinocyte cell lines and organ-cultured human skin.(26,27) HSP27 is expressed predominantly in the suprabasal epidermis in human skin,(28) whereas HSP70 predominates in the dermis compared with the epidermis.
These differences in location between HSP27 and HSP70 might explain the different time course of induction of these stress proteins upon O3 exposure. Interestingly, O3 induction of HO-1 showed a delayed time course compared with that for HSP27 and 70, in line with a previous study, which showed a peak of HO-1 induction at 18–24 h in rat lungs after O3 treatment.(29) It is therefore possible that bioactive compounds generated by products of O3 exposure may be responsible for the induction of HO-1 as was also shown after UV irradiation.(30,31) As HSPs are involved in cell proliferation, apoptosis and inflammatory response, O3-mediated HSPs induction can affect normal skin physiology. Thus, HSPs might provide an adaptive cellular response to O3; enhancing the expression of HSPs might turn out to be a new way to deal with the immediate and long-term consequences of O3 exposure. A prerequisite for the utilization of this concept is the development of nontoxic HSP inducers and their evaluation for clinical efficacy and safety. Furthermore, increased levels of metalloproteinase-9 (MMP-9; mRNA and activity) was observed after O3 exposure

) MMPs have been associated with the degradation of the basal membrane and play important roles in wound healing and in tumour development. In addition, MMPs may contribute to the enhancement of skin ageing and formation of wrinkles.(32) O3 is also able to modulate proliferative responses in mouse skin.(23) Proliferating cellular nuclear antigen (PCNA) is a protein identified as the polymerase-associated protein synthesized in the early G1 and S phases of the cell cycle involved in DNA replication and repair. PCNA is induced by stress responses that cause DNA damage;(33) it has been reported that PCNA gene expression can be induced in the lungs by diesel exhaust particles, another form of oxidative lung damage,(34) suggesting that oxidation can affect proliferative behaviour in target tissues.
O3 exposure can also affect cell differentiation. In skin tissue, we detected an increase of keratin 10 (K10) production after O3 treatment;23 K10 is a keratin produced in well differentiated, suprabasal keratinocytes; O3-induced changes in K10 suggest that O3, induces keratinocyte proliferation and differentiation.(35) It is not clear how O3 displays its effects, but recent studies have shown that it is able to induce the activation of the transcription factor, NF-KB, by phosphorylation of the kinase, IKBalfa-gama.(23)
Changes in the redox state have been shown to activate the NF-jB intracellular signalling pathway; this cascade includes several kinases and transcription factors. NF-jB-mediated signal transduction has been implicated in the regulation of viral replication, autoimmune diseases, tumorigenesis and apoptosis, and in the inflammatory response. In this regard, the activation of NF-KB is known to play a crucial role in COX-2 gene activation,(36) suggesting that O3 plays a role in the expression of numerous proinflammatory and adaptive inflammatory responses. It is not surprising that exposure of the skin to O3 can trigger several biochemical pathways leading to inflammation and affecting skin biology. On the other hand basic and clinical work developed during the last 15 years has shown that transient treatment and small O3 doses can reactivate useful body functions and might display therapeutic activity.(37)
Topical application

Interestingly, in spite of its instability, the O3 molecule can be stabilized as an ozonide between the double bonds of a monounsaturated fatty acid such as oleic acid.37 As a consequence, ozonated olive oil remains stable for 2 years at 4 Celsius. This preparation is proving to be ideal for the topical use of O3 in the treatment of chronically infected cutaneous and mucosal areas of the body. O3 is widely recognized as one of the best bactericidal, antiviral and antifungal agents and therefore it is profitably and practically employed as ozonated olive oil with well defined peroxide contents. The ozonated oil is now used topically for the treatment of war wounds, anaerobic infections, herpetic infections (HHV I and II), trophic ulcers and burns, cellulitis, abscesses, anal fissures, decubitus ulcers (bed sores), fistulae, fungal diseases, furunculosis, gingivitis and vulvovaginitis.(41) Matsumoto et al. tested the efficacy of the ozonated oil in the treatment of fistulae and chronic surgical wounds and, in a series of (28) patients, the ozonated oil was fully effective in 27 cases without side-effects.(42) Even radiodermatitis lesions in patients with cancer have been found to be beneficially influenced by exposure to O3(43) but far better results could be achieved with the simple application of ozonated oil.
Conclusions
Biological and clinical studies on the effects of O3 on the skin have shown that O3 can be either toxic, or safe at the point of use as a real drug, depending upon its dosage, length of exposure and the antioxidant capacity of the tissue exposed. The ambivalent character of O3 has been likened to the Latin god Janus;(44) indeed O3 is useful in the stratosphere but is toxic in the troposphere because of its chronic effects on the respiratory system, skin and mucosae.(45)
On the other hand, it has recently been observed that olive oil, which during ozonation traps O3 in the form of a stable ozonide, when applied to all sorts of acute and chronic cutaneous infections, slowly release O3 which, in comparison with conventional creams, displays effective disinfectant and stimulatory activities that lead to rapid healing. The dual behaviour of O3 fits well the concept of ‘hormesis’ that says the exposure of a living organism to a very low level of an agent harmful at high or chronic levels induces an adaptive and beneficial response.(46,47)
2005 British Association of Dermatologists • British Journal of Dermatology 2005





 HSP27 showed the earliest (2 h) and highest (20-fold) response to O3 compared with the delayed induction (12 h) of HSP70 and HO-1. Increased expression of HSP27 has been demonstrated following heating of both keratinocyte cell lines and organ-cultured human skin.(26,27) HSP27 is expressed predominantly in the suprabasal epidermis in human skin,(28) whereas HSP70 predominates in the dermis compared with the epidermis.
HSP27 showed the earliest (2 h) and highest (20-fold) response to O3 compared with the delayed induction (12 h) of HSP70 and HO-1. Increased expression of HSP27 has been demonstrated following heating of both keratinocyte cell lines and organ-cultured human skin.(26,27) HSP27 is expressed predominantly in the suprabasal epidermis in human skin,(28) whereas HSP70 predominates in the dermis compared with the epidermis.







